Acid Phenol Alcohol
Organic Chemicals 2025-03-02
Definition & Structure
Phenolic Alcohols are aromatic compounds featuring a hydroxyl (–OH) group directly bonded to a benzene ring.
- General Formula: Ar–OH (Ar = aryl group, e.g., C₆H₅)
- Key Feature: Exhibit both alcohol-like (weak acidity) and aromatic properties.
Classification & Examples
| Type | Example | Structure | Key Applications |
|---|---|---|---|
| Simple Phenols | Phenol (C₆H₅OH) |  |
Disinfectants, resin production |
| Polyphenols | Catechol (C₆H₄(OH)₂) | 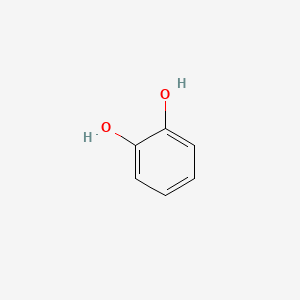 |
Antioxidants, dyes |
| Bisphenols | Bisphenol A (BPA) | 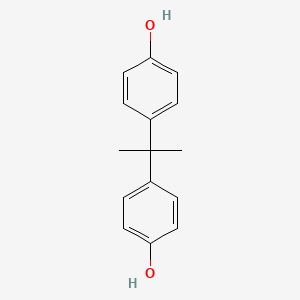 |
Polycarbonate plastics |
| Thymol/Menthol | Thymol (C₁₀H₁₄O) | 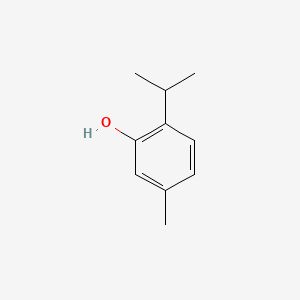 |
Antiseptic, flavoring |
Key Properties
| Property | Behavior | Industrial Relevance |
|---|---|---|
| Acidity | Weakly acidic (pKa ~10) | Forms phenoxide salts (e.g., C₆H₅O⁻Na⁺) |
| Antimicrobial | Disrupts cell membranes | Used in disinfectants (e.g., Lysol®) |
| Solubility | Water-soluble (small phenols), lipid-soluble (larger derivatives) | Formulation flexibility |
Industrial Applications
| Industry | Use Case | Example Compound |
|---|---|---|
| Pharmaceuticals | Antiseptics, analgesics | Salicylic acid (precursor to aspirin) |
| Cosmetics | Preservatives, fragrances | Eugenol (clove oil) |
| Polymers | Epoxy resins, adhesives | Bisphenol A (BPA) |
| Agriculture | Pesticides, herbicides | 2,4-Dichlorophenoxyacetic acid (2,4-D) |
Disclaimer: The above content is for reference and communication only among industry insiders, and does not guarantee its accuracy or completeness. According to relevant laws and regulations and the regulations of this website, units or individuals who purchase related items should obtain valid qualifications and qualification conditions.



